Supporting Registries Reporting MIPS Measures in 2021
As healthcare leaders know, the process of reporting quality measures for the Merit-Based Incentive Program (MIPS) grows more complex and time-intensive every performance year.
Participating in MIPS gives eligible clinicians the opportunity to earn higher payments for providing quality, cost-effective care. For many, reporting MIPS data to CMS means participating in a clinical data registry.
What is a Clinical Data Registry?
A clinical data registry, also known as patient registry or disease registry, is a database that collects healthcare information for a population with a particular disease or condition to understand how well various treatment and outcomes work.
Types of MIPS Registries Include:
- Qualified Clinical Data Registry (QCDR) are specialized, CMS-approved registries used to collect and report MIPS data
- Qualified Registries (QR) also collect and report data to CMS. The main difference is that QCDRs can collect and report specialty-specific measures (that are not part of the MIPS measure set) and are often more useful for specialist physicians.
The 2021 MIPS Program and Final Rule
The 2021 CMS Final Rule puts an increased emphasis on data validation for QDCRs and qualified registries.* QCDRs/QRs reporting MIPS data in 2021 (for payment in 2023) must conduct data validation audits, with specific requirements, on an annual basis.
2021 registry requirements include:
- Validating data submitted for each performance category and each submitter type
- Reviewing clinical documentation to validate the action or outcome measured
- Increasing the sample size of randomized audits to three percent of TIN/NPIs reporting data (maximum of 50 TIN/NPIs)
How Can Telligen Help?
Registries reporting data to MIPS in 2021 need to have a solid data validation plan ready.
How does Telligen support registries?
Download the brochure to learn more about our registry solutions.

For QCDRs and QRs, Telligen offers client-specific auditing and validation plans that transform complicated, resource-intensive tasks into streamlined processes that maximize revenue while ensuring quality data is established to guide research and improvement efforts.
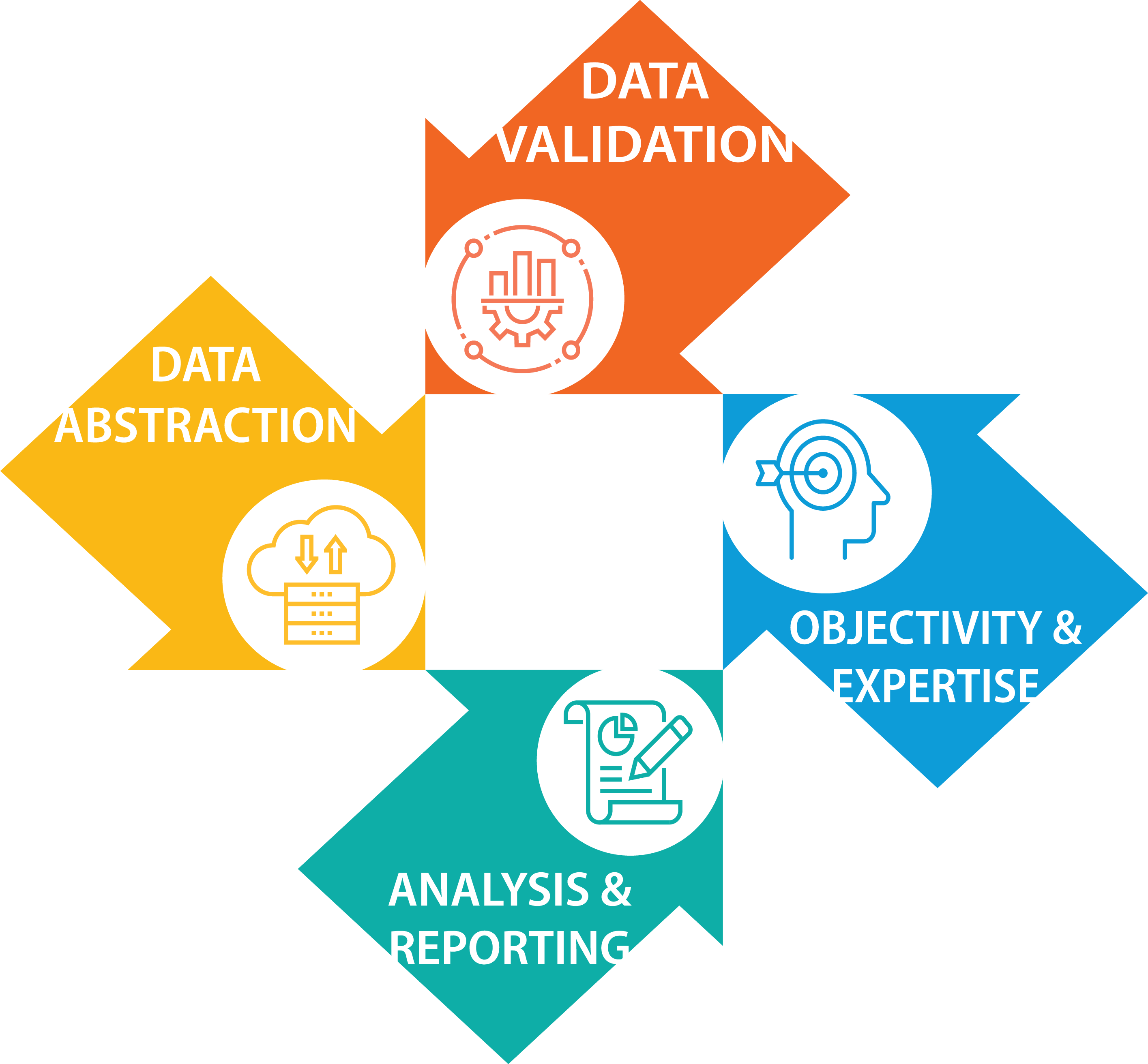
Our specialized services and support for registries include:
- Data Collection: Collect data using secure file transfer protocols & remote abstraction tools in compliance with client guidelines, HIPAA regulations & government regulations
- Data Validation: Validate the quality (consistency, reliability and validity) of abstracted data to ensure clinical documentation supports the performance reported
- Expertise & Objectivity: Ensure an objective, unbiased validation approach led by experts with extensive backgrounds in medical coding & hands-on data abstraction
- Analysis & Reporting: Develop a comprehensive final report that calculates an element-specific & overall accuracy score
*Specific to data validation policy, §414.1400 (b)(2)(iv) & §414.1400(c)(2)(iii)


